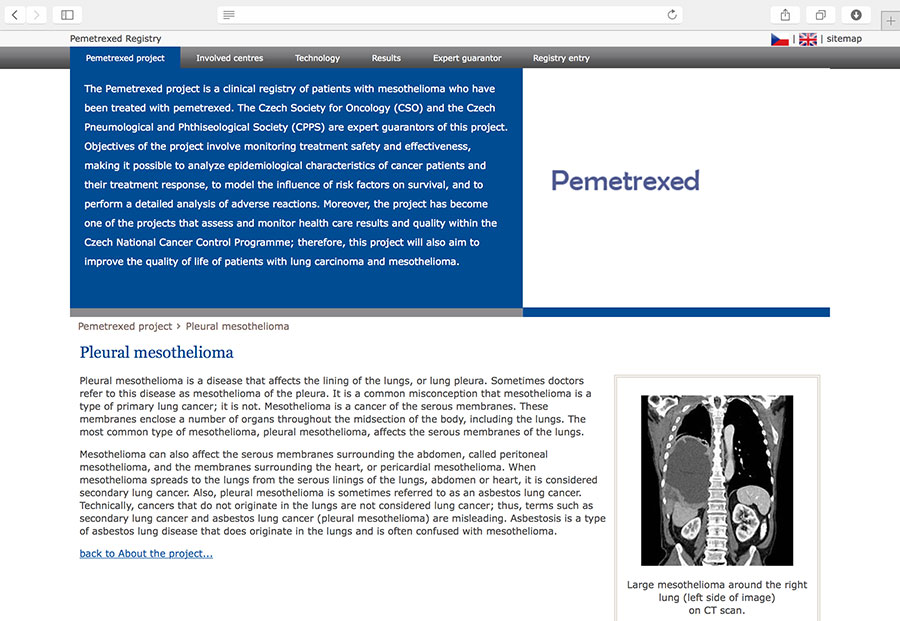
Clinical registry of pleural mesothelioma patients who have been treated with pemetrexed (Alimta®).
start of the project: 09/2009
end of the project: 07/2011
The Pemetrexed project is a clinical registry of patients with non-small cell lung cancer (NSCLC) or mesothelioma who have been treated with pemetrexed (Alimta®). The Czech Society for Oncology (CSO) and the Czech Pneumological and Phthiseological Society (CPPS) are expert guarantors of this project. Objectives of the project involve monitoring treatment safety and effectiveness, making it possible to analyze epidemiological characteristics of cancer patients and their treatment response, to model the influence of risk factors on survival, and to perform a detailed analysis of adverse reactions. Moreover, the project has become one of the projects which assess and monitor health care results and quality within the Czech National Cancer Control Programme; therefore, this new project will also aim to improve the quality of life of patients with lung carcinoma and mesothelioma, and to make the best use of available resources for these patients' treatment.
The project was initiated at the Institute of Biostatistics and Analyses of the Masaryk University (IBA MU) and has continued at the Institute of Biostatistics and Analyses Ltd (IBA Ltd), a university company.
Note: Data collection for the NSCLC part of the Pemetrexed registry was terminated in July 2011. Data on pemetrexed administration to NSCLC patients are currently being entered into the Tulung registry, while data on pemetrexed administration to patients with MPM (malignant pleural mesothelioma) continue to be entered into the Pemetrexed registry.
- Expert guarantor: Prof. Vítězslav Kolek, M.D., Ph.D.
- Partners: 10 specialized centres in the Czech Republic
- Anaesthesiology, intensive care medicine
- Cardiology, angiology
- Diabetology, dietetics
- Educational projects
- Emergency medicine, toxicology
- Endocrinology, metabolism
- Gastroenterology, hepatology
- Haematology, haematological oncology
- Immunology, allergology
- Medical genetics
- Nephrology
- Neurology, neurosurgery
- Nursing care
- Obstetrics, gynaecology
- Oncology, radiotherapy
- Ophthalmology
- Paediatrics, neonatology
- Pathology, laboratory medicine
- Pulmonary medicine
- Rheumatology, physiotherapy
- Screening programmes
- Surgery, traumatology, orthopaedics
- Urology